Crystalline and Amorphous Solid State
Solid State
The solid-state (or solid phase) is the state of matter characterized by having a definite shape and volume.
This specific property of the solid-state arises from the presence of specific bonding and non-bonding interactions between the atoms within the solid phase. These interactions are strong enough to constrain the atoms and/or molecules within specific physical spaces.
If the atoms are arranged in an ordered manner throughout the solid (long-range order), it is referred to as a crystalline solid.
Crystal, from the Greek "kristallos", literally means frozen water, ice.
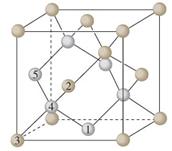
Crystals are characterized by having a specific repeating unit, called the unit cell, which contains a certain number of atoms constrained in specific spatial positions.

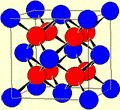
The unit cell is defined by the unit cell parameters:
Axes a, b, c
Angles α, β, γ
α: Opposite to a, the angle between axes b and c
β: Opposite to b, the angle between axes a and c
γ: Opposite to c, the angle between axes a and b
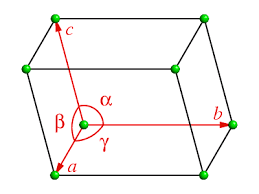
The direction of the three crystallographic axes is chosen to define a right-handed system.
The unit cell is bounded by:
6 faces, grouped into pairs:
- 2 A faces, opposite to a, defined by axes b and c
- 2 B faces, opposite to b, defined by axes a and c
- 2 C faces, opposite to c, defined by axes a and b
12 edges, grouped into quartets:
- 4 equivalent edges for each axis a, b, and c
8 corners, all equivalent.
The volume V of the unit cell is given by:
V = a·(b x c)
The scalar product of one axis with the vector cross product of the other two axes.
V = a*b*c*√(1-cos2α-cos2β-cos2γ+2cosα∗cosβ∗cosγ)
In orthogonal systems (α=β=γ=90°) the expression simplifies to:
V=a*b*c.
In the crystal, this unit cell repeats identically by translation along the three spatial directions.
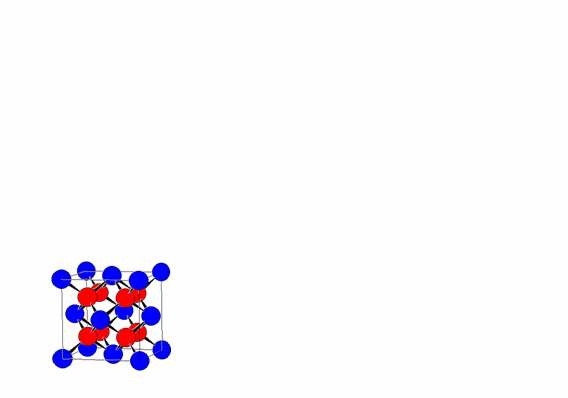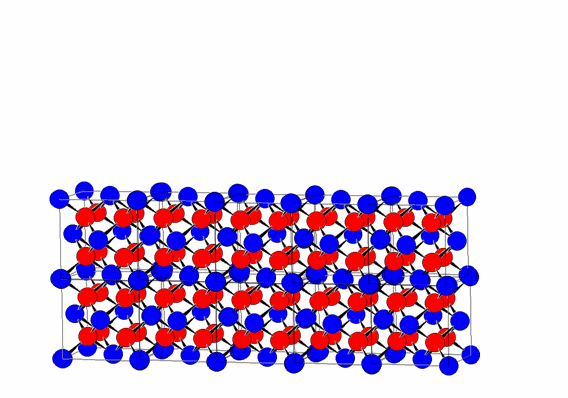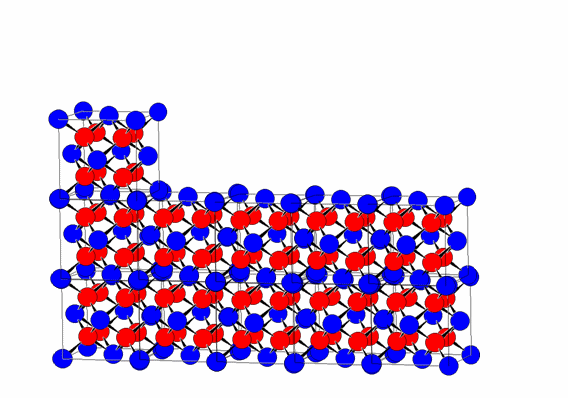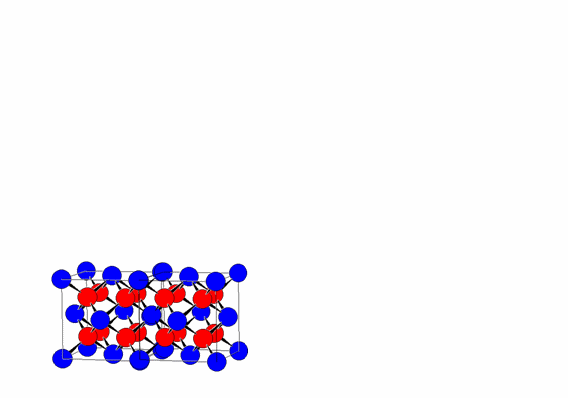
This regular repetition of the unit cell by translation along the three directions a, b, and c is one of the fundamental characteristics of crystalline materials, distinguishing them from other forms of condensed matter, such as liquids and glasses.
The periodic translation of the unit cell (translational symmetry) generates the crystal lattice.
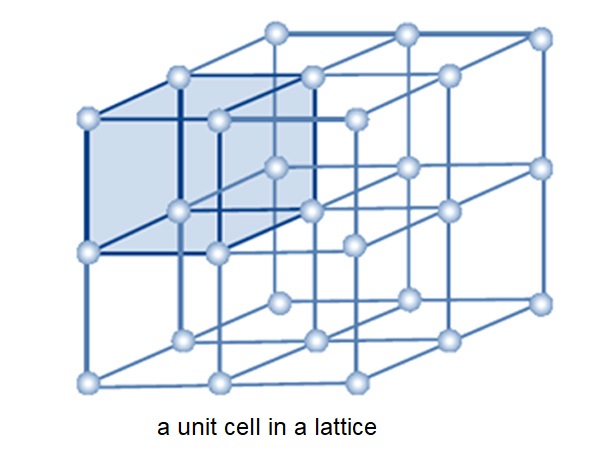
The vertices of the unit cells define the lattice nodes, which are all equivalent due to the translational symmetry of the lattice (assuming an infinite lattice).
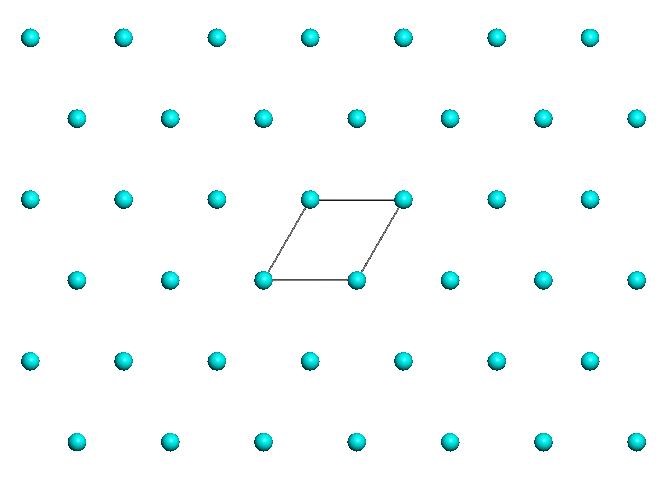
Lattices can be one-dimensional (1D), two-dimensional (2D), or three-dimensional (3D), depending on whether the periodicity is observed along a line, on a plane, or in three-dimensional space.
For an infinite three-dimensional lattice, choosing a node, O as origin, and a set of primitive vectors a, b, and c, each node of the lattice is uniquely defined by the vector:
The choice of the lattice origin and the specific location of the node is completely arbitrary to reconstruct the entire content of the crystal by translation, although there are conventions in crystallography. For example, one convention is to choose a symmetry element, such as an inversion center, as the origin of the unit cell. By applying the translational properties of the unit cell to this vertex, the nodes of the entire crystal lattice are generated. It should be noted that, if the origin is placed on a symmetry element, considering that all nodes are equivalent to each other, all nodes will have this symmetry element.
Analysis of the crystal structure using X-ray diffraction

X-ray diffraction is the primary analytical technique for determining crystalline structures. Determining the crystal structure provides insight into the molecular geometry (structural parameters such as bond distances, bond angles, and torsion angles) and the characteristics of covalent, ionic, and/or intermolecular interactions present in the solid state.
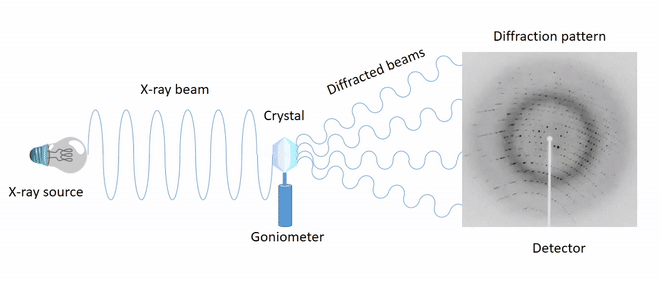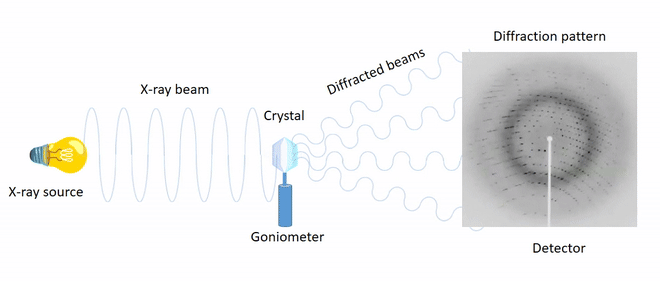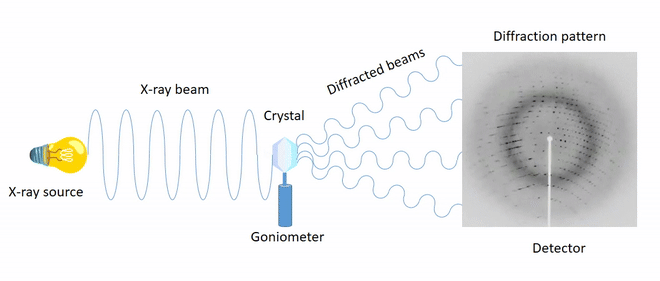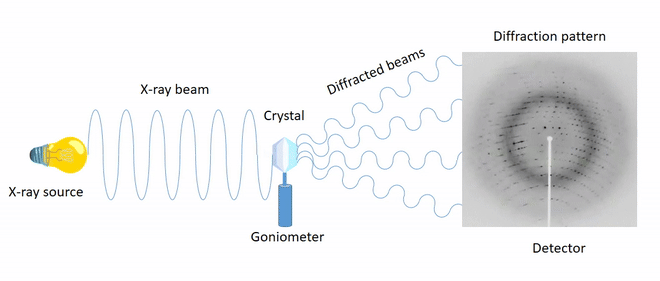
The directions of the diffracted beams (diffraction geometry) depend on the unit cell parameters, while the intensities of the spots depend on the positions of the atoms in the cell.
Processing diffraction spectra makes it possible to build electron density maps, enabling the identification of the regular arrangement of atoms within the crystal.
Understanding the crystal and molecular structure is fundamental for studying the behavior of both inorganic and organic materials, and biological systems, such as proteins.
Amorphous and Crystalline Solids
If the atoms are arranged in a way that lacks a repetitive order, the solid is referred to as an amorphous solid. In this case, the atoms maintain only short-range order.
On the contrary, crystalline solids are characterized by having both short-range and long-range translational order.
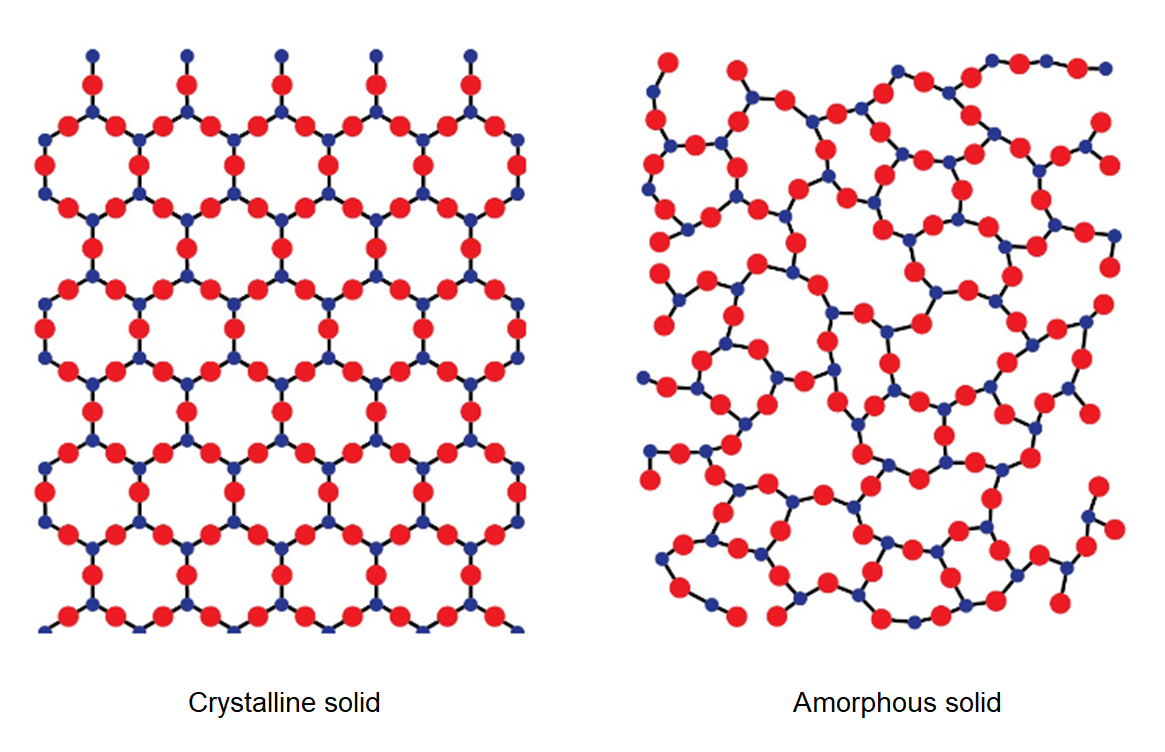
Difference between Quartz and Glass
Common glass has high hardness, but unlike a quartz crystal, it fractures irregularly. This is because glass consists of SiO₂ units arranged randomly, without the regular repetition of atoms found in quartz crystals.
In glasses, the disordered atomic arrangement characteristic of the molten state remains "frozen". Glasses are also considered highly viscous supercooled liquids, which can still flow, although at an extremely slow and practically unobservable rate.
The cohesive forces in solids determine the type of crystals.
COVALENT BOND > COVALENT CRYSTALS
 [Diamante]
[Diamante]
The covalent bond is directional, and atoms in covalent crystals generally exhibit a low coordination number. The density is relatively low compared to metals, and covalent crystals are hard and difficult to cut. They also have insulating (or semiconducting) properties.
Atoms have a high bond energy, and the covalent crystals feature a high melting point with low expansion and compression coefficients (thermoelastic properties).
IONIC BOND > IONIC CRYSTALS
 [ NaCl]
[ NaCl] 
The ionic bond is non-directional, and ions exhibit high coordination numbers. The density of ionic crystals is high and they are hard but brittle and have insulating (or semiconducting) properties.
Ionic crystals have a high lattice energy, a high melting point, and low expansion and compression coefficients (thermoelastic properties).
METAL BOND > METAL CRYSTALS
The electrons in the conduction band are delocalized throughout the solid, the bond is non-directional, and atoms exhibit high coordination numbers. The metal density is high, the solid is malleable, and it has conductive properties. Metals generally show relatively high thermal expansion coefficients, with characteristics of shininess and medium-to-low melting points.
NON-BONDING INTERACTIONS > MOLECULAR CRYSTALS
 [Zucchero]
[Zucchero]
The molecules, characterized by directional covalent bonds, are held together by non-bonding van der Waals interactions and hydrogen bonds. The density of the molecular crystals is low. Molecular crystals generally melt at low temperatures, and exhibit high expansion and compression coefficients (thermoelastic properties).

 [Au]
[Au]