Structure of Inorganic Solids
Structure of Inorganic Solids
The most direct way to describe crystalline structures, which provides all the necessary information, is to specify the unit cell (cell parameters), the symmetry (space group), and the positions of the atoms within the asymmetric unit (fractional/crystallographic coordinates), and, if applicable, the thermal motion of the atoms (crystallographic description). In terms of atom positions, the entire crystal structure can be reconstructed from the cell parameters, the coordinates of the independent atoms within the cell, and the symmetry. However, neither the description of the independent unit alone nor that of the entire cell often gives a clear or satisfactory image of the three-dimensional structure, particularly in the case of non-molecular inorganic compounds. There is a problem both in visualizing and rationalizing the structure.
For example, the crystallographically independent unit of the alumino-silicate framework of the zeolite Ca-chabazite (Ca[Al₂Si₄O₁₂]·X H₂O) shown below, while exhaustive in terms of structural information, fails, through its graphical representation, to fully convey the structural properties of this molecular sieve.
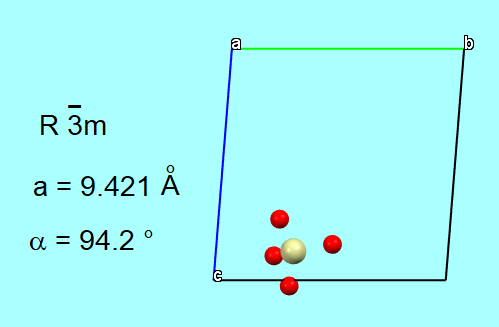
and neither does the representation of the entire unit cell
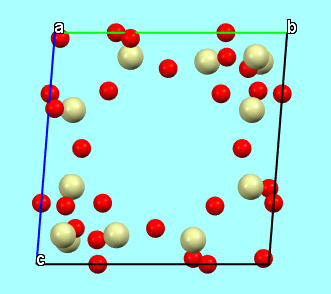
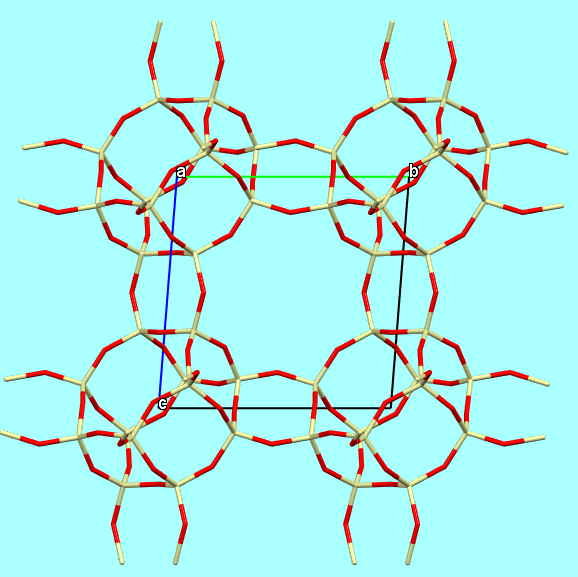
Structures, in general, can be represented and illustrated in various ways, depending on the aspects one wishes to highlight. For example, the framework structure of chabazite can be illustrated through:
a) a “sticks” model, if the goal is to emphasize the atoms connected by covalent bonds
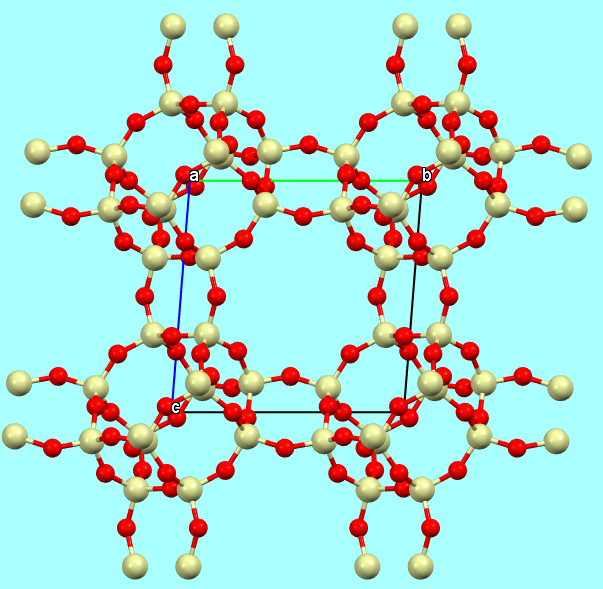
b) a “ball and sticks” model, if the aim is to highlight not only the bonds but also the positions of the atoms/nuclei;
c) a model with thermal ellipsoids, if the goal is to highlight the thermal motion of the atoms within the crystal structure;
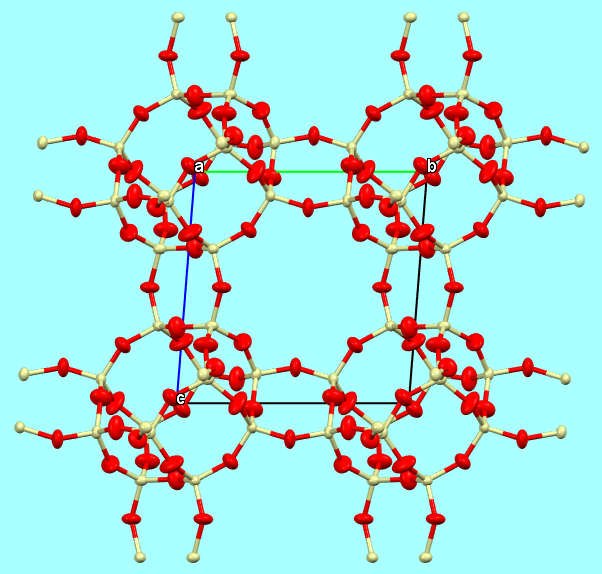
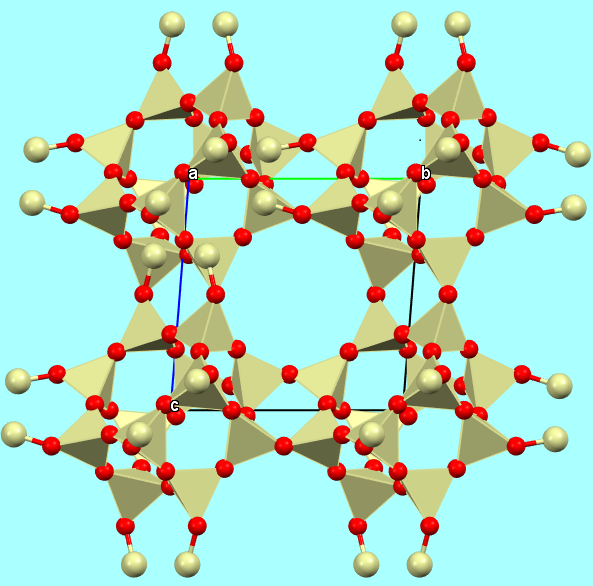
d) a model with coordination polyhedra, to highlight the coordination geometry and the connections between the various polyhedra;
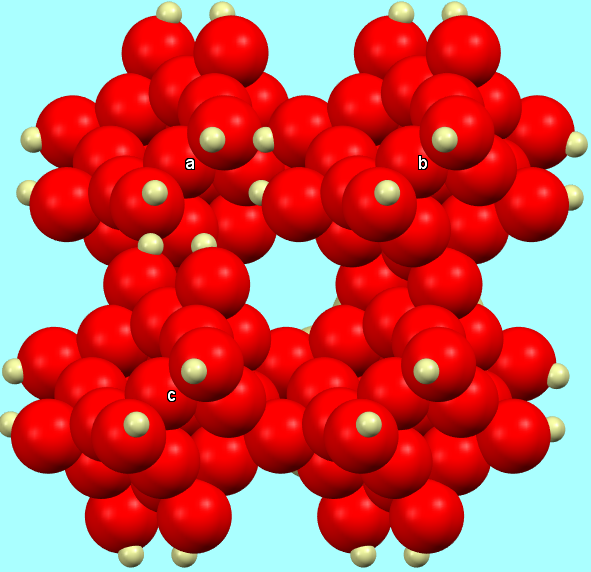
e) a “space-fill” model, to emphasize the spatial occupation of the system and the close packing of spheres.
Chabazite-Ca framework
Chabazite-Ca full structure
Geometric and Topological Aspects
We can identify three main approaches to describe the crystal structure of inorganic solids:
-
Sphere packing, an approach suitable for studying systems characterized by isotropic interactions with spherical symmetry, such as metallic and ionic systems, where high coordination numbers are typically present;
-
Polyhedral organization, an approach mainly suited for ionic systems with intermediate coordination numbers (neither too high nor too low);
-
2D and 3D networks, an approach appropriate for systems with directional covalent bonds, which generally exhibit low coordination numbers.
Reference Structures
A crystal structure is often discussed and compared with well-known structures (reference structures). Therefore, the systematic study of reference crystal structures that represent specific types of crystal packing is of considerable importance.
For example, the analysis of sphere packing in simple compounds with the formula AmXn, where the X atoms are closely packed and the A atoms occupy tetrahedral or octahedral sites, has revealed interesting constraints related to coordination geometry. It has been shown that many crystal structures represent a compromise between conflicting requirements of packing atoms of different types, sizes, and coordination preferences. Considering the local environment of individual ions, the energetically preferred coordination polyhedron for an ion may be geometrically impossible in a given 3D structure.
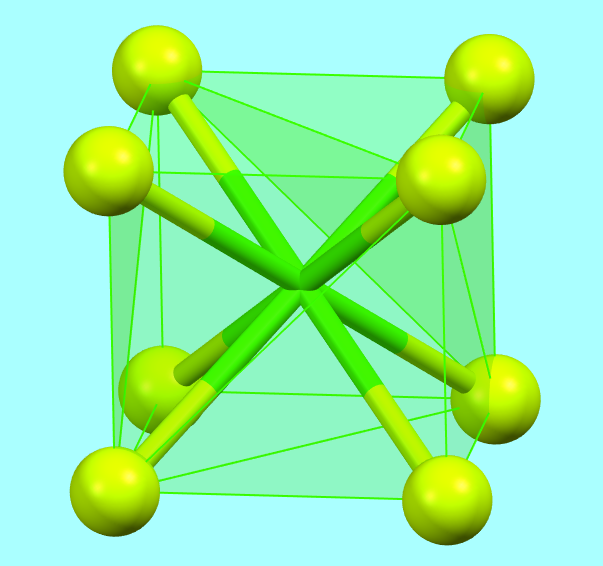
For instance, in the ionic structure of fluorite (CaF₂), calcium ions exhibit cubic 8-fold coordination rather than square antiprismatic or dodecahedral coordination (such as the triangular dodecahedron or scalenohedral disphenoid), which would be more favorable from an energetic point of view. However, these two types of 8-fold coordination polyhedra are incompatible with AX₂-type structures.
Fluorite, CaF2
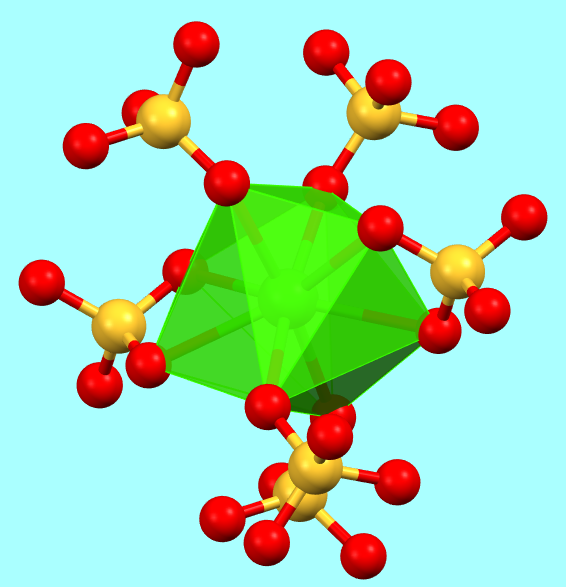
They do occur in structures where the geometric constraints of crystal packing are less strict due to the presence of non-spherical molecular ions, as seen in calcium sulfate (CaSO4).
Calcium sulfate, CaSO4
While some structures are found almost exclusively in systems with a particular type of bonding (for example, the rutile structure of TiO₂ is a reference structure for essentially ionic compounds of the AX₂ type), other simple structures having a specific type of bonding are not necessarily indicative of such a bond. For instance, the NaCl reference structure is adopted not only by ionic oxides and halides, but also by compounds, such as nitrides and carbides of transition metals, and certain intermetallic compounds.
It is therefore preferable, in general, to describe simple AmXn-type structures as geometric entities with specific topological properties rather than as strictly ionic, covalent, or metallic structures.
